Authors: Lance Ramsey, Ellen Gualtieri, Eric Zhao
How can Fluorescence Recovery After Photobleaching (FRAP) save you time?
FRAP can be used to determine if a particular crystallization setup has the potential to crystallize. Scientists can check protein constructs, ligands, LCP host lipids and precipitants all in just a 30 minute FRAP assay. Those conditions that show mobility have the potential to then successfully crystallize.
protein diffusion in LCP = potential to crystallize
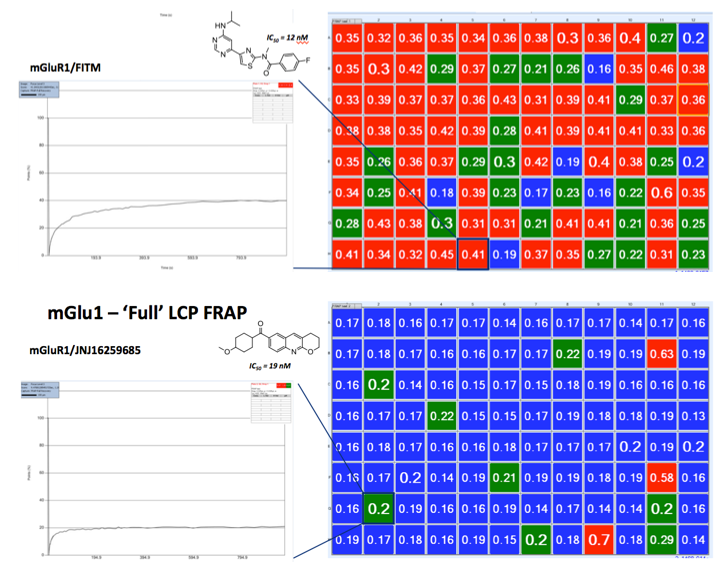
Prescreening Ligands with FRAP
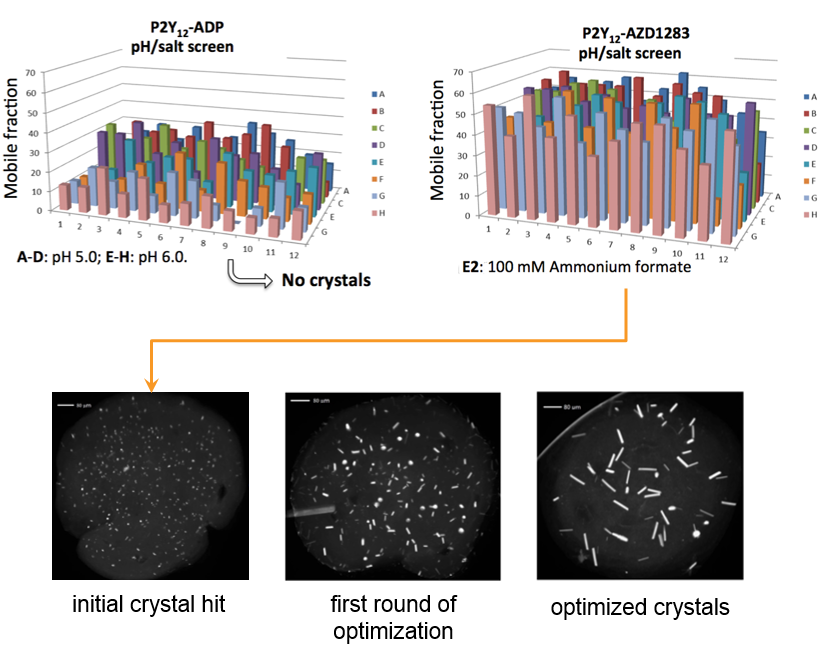
Determination of the best ligand for crystallization trials are assessed with High Throughput LCP-FRAP experiments. Receptor complex of purinergic P2Y12with AZD1283 showed much higher diffusion in LCP than with the ADP complex. Only the P2Y12-AZD1283 complex generated crystals after extensive screening of both complexes, demonstrating a strong correlation between mobility and crystallization.1
Optimizing Serotonin Receptor Subtypes with HT LCP-FRAP
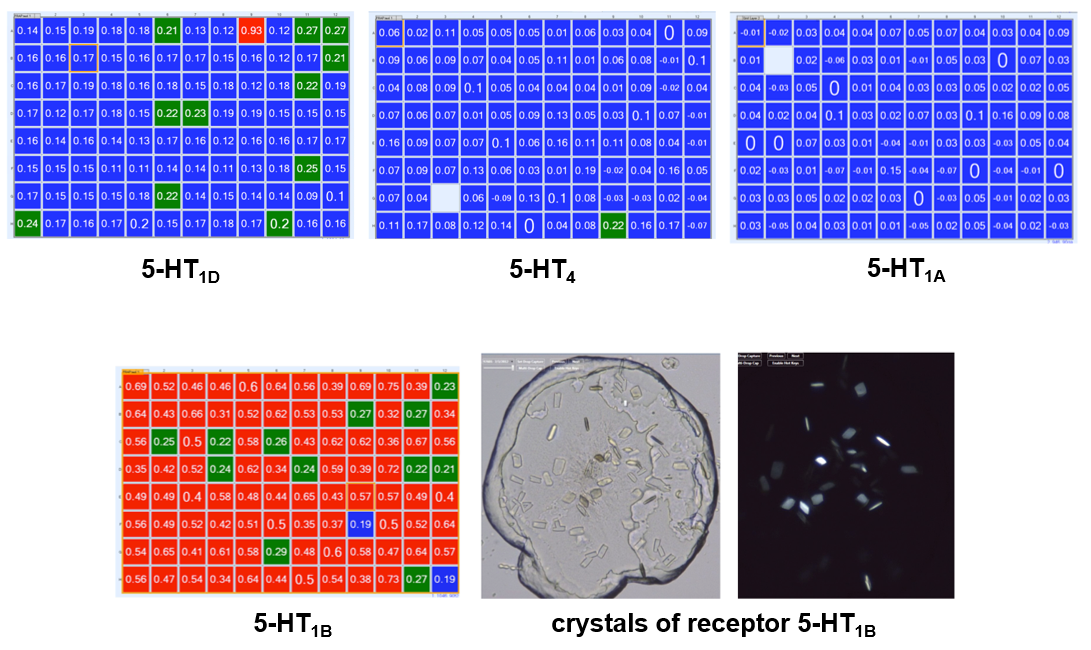
High Throughput LCP-FRAP experiments are performed to determine the optimal serotonin (5-HT) receptor subtype for crystallization trials. Four representative results for varying receptor subtypes are shown in a color coded matrix where blue highlights wells with little to no mobility, green indicates moderate mobility and red identifies high mobility and highest probability for crystallization. 5-HT1Breceptor was the only protein to show diffusion in LCP and yielded crystals that were used for structure determination.1
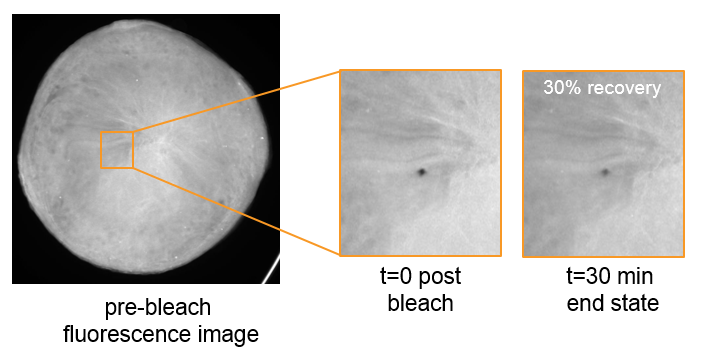
Full Recovery FRAP Analysis
Full Recovery FRAP analysis provides the ability to probe a single well condition to determine diffusion rates. The automated FRAP imager from FORMULATRIX® measures the fluorescence recovery on the same spot in defined intervals for a 20-30 minute period. The full recovery curve gives information regarding lipid diffusion and heterogeneity of the sample, helping to rule out false positives.1
How it Works
Screen 96 conditions in 30 minutes
FRAP automatically analyzes 96 well LCP plates to determine percent mobile fraction for each drop. Each well is bleached by a focused laser spot, and the recovery of fluorescence after a fixed time point is measured. The percentage of proteins that are mobile in the drop are indicative of whether or not crystallization may occur.
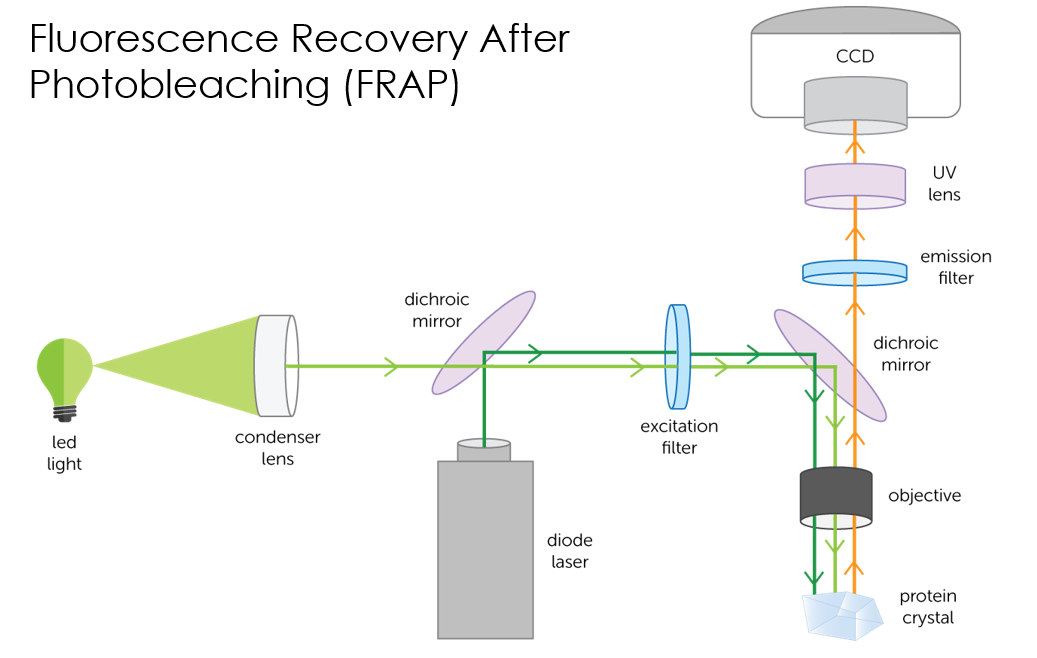
The FRAP imager emits a photobleaching laser pulse which bounces off a dichroic mirror, through an excitation filter, and off a 2nd dichroic mirror, bleaching the sample.
A LED shines light through a condenser and the light follows the same path as the laser, illuminating the sample.
Fluorescent images are then processed.
Reference
1Fenalti, G., Abola, E., Wang, C., Wu, B., Cherezov, V. Fluorescence Recovery After Photobleaching in Lipidic Cubic Phase (LCP-FRAP): A Precrystallization Assay for Membrane Proteins. In: Arun Shukla, editor, Methods in Enzymology,Vol. 557, Burlington: Academic Press, 2015 pp. 417-437.